Faster, more efficient CRISPR editing in mice
Giving embryos a jolt of electricity drives CRISPR-Cas9 into cells for more efficient gene editing
May 27, 2016
UC Berkeley scientists have developed a quicker and more efficient method to alter the genes of mice with CRISPR-Cas9, simplifying a procedure growing in popularity because of the ease of using the new gene-editing tool.
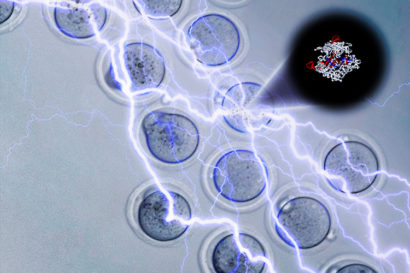
Electroporation jolts embryos with electricity to open holes that allow the CRISPR-Cas9 molecule (upper right) to enter and edit the DNA. Andrew Modzelewski, Lin He image.
While CRISPR-Cas9 has drawn worldwide attention because of its potential to correct simple hereditary diseases in humans, basic researchers are excited about its ability to help them understand the causes and develop treatments for more complex diseases, including cancer and dementia.
To do that, they need to knock out or modify specific genes in lab animals — in particular, mice — and see what goes awry. Today’s gold standard for creating these “knockouts” or “knockins” is to edit the genes inside mouse embryonic stem cells, use these cells to create mosaic mice, and then crossbreed the mice to get a pure genetic strain. Because of CRISPR-Cas9’s ability to precisely alter or replace genes, the editing is increasingly being done directly in the fertilized egg, or early embryo.
The new method, called CRISPR-EZ (CRISPR RNP electroporation of zygotes), makes genome editing in mouse embryos even easier.
“The key fundamental insights about the biological significance of a gene usually come from in vivo gene-editing studies, in which you generate mice with an altered gene,“ said lead researcher Lin He, a UC Berkeley associate professor of molecular and cell biology. “But it is a major committment to make a novel knockout with genome engineering. I think this technology could greatly reduce the technical barrier for this type of effort and will allow people to focus more on the science rather than be consumed by the process of genetically engineering mice.“
The new method, described in a paper in press at the Journal of Biological Chemistry, gets around a time-consuming bottleneck in creating knockout mice: using microscopic needles to inject gene-editing molecules into a fertilized egg.
The UC Berkeley researchers found that a simple lab technique called electroporation works much better, allowing them to insert CRISPR-Cas9 gene-editing molecules into embryos with nearly 100 percent success. Electroporation uses a jolt of electricity to create holes in the embryos through which molecules can enter.
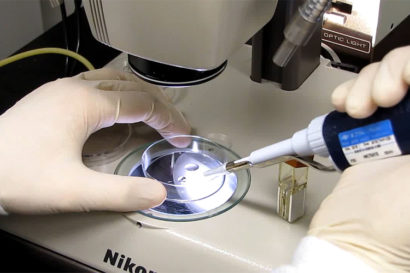
Using a micropipette, researchers suction up embryos in preparation for inserting CRISPR-Cas9 by electroporation. Lin He image.
Using CRISPR-EZ in a pilot experiment, He’s team successfully disrupted both copies of a target gene in 88 percent of the mice. The procedure generated a much greater number of edited mice compared to CRISPR microinjection, largely due to a significant improvement in embryo viability. CRISPR-EZ is a simple and cost-effective methodology, and can be performed on many embryos at once and takes only miliseconds, He said.
“In the not too distant past, it would cost at least $25,000 and take at least 6 months to make a knockout mouse,” said Russell Vance, a UC Berkeley professor of molecular and cell biology and director of the Cancer Research Laboratory, where the transgenic mouse work was performed. “With CRISPR, and improvements such as CRISPR-EZ, the costs and time have both dropped at least 10-fold. These technical innovations make the mouse an even more powerful tool for modeling human diseases.”
The UC Berkeley group is now working with several transgenic mouse facilities in hopes that they will adopt and improve this electroporation technique, which she suspects will also simplify the creation of other transgenic mammals.
Knockouts require IVF team
Creating transgenic mice requires a skilled in vitro fertilization team. Technicians use hormone injections to prepare the females for mating, after which they harvest the fertilized eggs and, before the eggs start to divide, inject them one at a time using a fine needle. Currently, technicians inject two RNA molecules – messenger RNA (mRNA), which codes for the Cas9 protein, and guide RNA, which provides the address for CRISPR-Cas9’s target – and hope that the mRNA is properly translated into Cas9 protein and that the protein correctly combines with guide RNA.
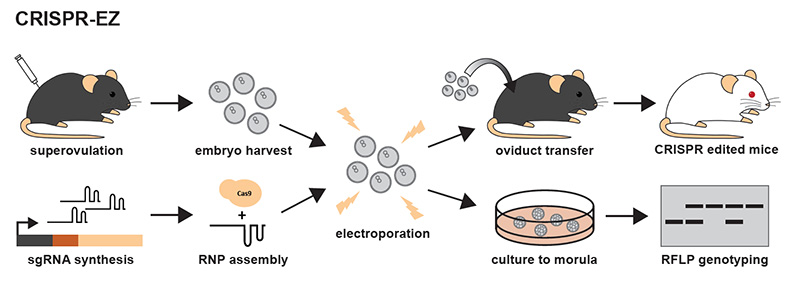
Creating transgenic mice involves harvesting fertilized eggs and electroporating them to insert the CRISPR-Cas9 molecule, then implanting the edited embryos in another mouse. The targeted gene is linked with a gene that produces white fur, making it easy to see which mice have been successfully edited.
The engineered embryos are implanted into a falsely pregnant mouse, where they gestate for about 20 days before birth. Given the inevitable embryo deaths during injection and the failure of the embryos to implant or go to term, the live-birth rate is low, He said.
“The actual percentage of live births from injected embryos is around 10 to 15 percent for most transgenic facilities, which is a problem with the procedure,“ she said. “Sometimes people collect more than 100 embryos just to generate one or two mice with desirable gene editing.“
Electroporation appears to do less damage to the embryos than microinjection: between 30 and 50 percent of the embryos resulted in live births.
Apparently, too, inserting pre-assembled CRISPR-Cas9 molecules into the fertilized egg is a more effective way to edit genes than injecting two molecules – the mRNA and guide RNA – and hoping that they properly self-assemble. Of the live births, 88 percent of the mice had both copies of the target gene edited – a higher success rate than usual for transgenic procedures, He said.
For a more complex procedure – modifying a short DNA sequence within a gene – the method was successful 42 percent of the time in a pilot experiment.
“With such a high success rate, you can use this to test your guide RNAs very quickly,“ she said. “If your CRISPR-EZ doesn’t work, it’s not because of delivery; it’s likely because your guide RNA design needs to be improved.“
The high embryo viability and very high gene-editing efficiency mean researchers need to use fewer mice and can conduct several transgenic experiments simultaneously.
Accidental discovery
In her lab, He studies small bits of RNA called microRNA, which modify how DNA is transcribed and thus control important processes inside a cell. Some types of cancer have been linked to problems with miRNA.

Using a micropipette, researchers suction up embryos in preparation for inserting CRISPR-Cas9 by electroporation. Lin He image.
Looking for a simpler way to create transgenic mice to study these regulatory processes, postdoctoral fellow Andrew Modzelewski tested electroporation to see if he could get eggs to more easily take up the Cas9 mRNA and the guide RNA. Despite other researchers’ apparent success with mRNA, his initial attempts were unsuccessful. One evening, finding himself out of mRNA and not wanting to waste the prepared embryos, he borrowed CRISPR-Cas9 protein from a neighboring lab, assembled RNP complexes and electoporated them instead.
“It has worked like magic ever since,” He said. “You would never think that this would work, because Cas9 is a gigantic molecule. I was surprised that such a huge protein could be electroporated efficiently.“
While most universities have transgenic labs where microinjections are performed, electroporation simplifies the procedure enough that individual labs might eventually do it themselves, she predicted.
Graduate student Sean Chen and postdoctoral fellow Andrew Modzelewski spearheaded this study, Other co-authors of the paper are undergraduate Benjamin Lee and Angus Yiu-Fai Lee, director of the Gene Targeting Facility in the Cancer Research Laboratory. The work also benefited from input from Jacob Corn, Mark DeWitt and Jennifer Doudna of UC Berkeley’s Innovative Genomics Initiative.
This work was supported by the National Cancer Institute (R01 CA139067) and National Institute of General Medical Science (RO1 1R01GM114414) of the National Institutes of Health.
RELATED INFORMATION