Engineers develop cancer-targeting nanoprobe sensors
UC Berkeley scientists have created smart nanoprobes that may one day be used in the battle against cancer to selectively seek out and destroy tumor cells, as well as report back on the mission's status.
January 29, 2010
Scientists at the University of California, Berkeley, have created smart nanoprobes that may one day be used in the battle against cancer to selectively seek out and destroy tumor cells, as well as report back on the mission’s status.
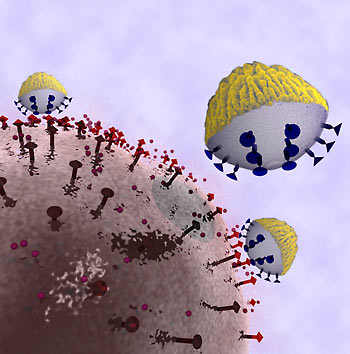
UC Berkeley scientists are designing smart nanoprobes, called nanocorals, to selectively attach to cancer cells, deliver therapeutic drugs and report on the local molecular environment. One side of the nanocorals is designed to selectively target the cell, while the other has a roughened surface to sense tell-tale chemical particles in the environment. (Benjamin Ross and Liz Wu, UC Berkeley)
A small number of research teams around the world have been developing target-specific nanoprobes for the past 10 years in an effort to reduce — and perhaps eliminate — the toxic toll chemotherapy takes on the healthy cells that reside near their diseased counterparts.
What had been missing, however, is a mechanism by which the nanoprobes could not only find the cancer cell, but also relay information once they latched onto the target. The UC Berkeley team created such multi-functioning probes, which they have dubbed nanocorals.
The development of the new nanocorals is the cover story for the Feb. 22 print issue of the peer-reviewed journal Small.
“If you’re sending a satellite into space, you need it to do more than one thing. It must reach its target, detect its surroundings, and communicate back to ground control,” said Luke Lee, Lloyd Distinguished Professor of Bioengineering at UC Berkeley and head of the UC Berkeley team that developed the nanocoral. “The same is true in the molecular galaxy. We need probes that can find a diseased cell, treat it, and tell us about the local environment so we can determine whether the treatment is working. The nanocoral probes we invented are an important step in this direction.”
The tiny probes measure a few hundred nanometers in diameter — one-thousandth the width of a human hair, and one-hundredth the size of most cancer cells. The team’s insight was to combine different materials — roughened gold on one side, and smooth polystyrene on the other — onto a single probe.
The name of the new probe is inspired by natural sea corals, which use rough surfaces to enhance the capture of light and food particles.
“Like natural corals, the highly roughened nanocoral surface is designed to capture molecules near the probes, and report their presence back to researchers,” said Benjamin Ross, a Ph.D. student in UC Berkeley’s Applied Science and Technology Program, and one of two co-lead authors of the study. “The type of molecules present — or absent — at the cell’s surface can provide telltale signs of how a cell is reacting to the new drug being delivered.”
The sensing side of the nanocoral relies upon a technique called surface-enhanced Raman spectroscopy (SERS), which takes advantage of the electromagnetic excitations that occur as molecules make contact with the roughened surface of a metal, such as gold. Molecules produce oscillations that resonate at signature frequencies when exposed to laser light, revealing their presence to the scientists.
The researchers verified the sensitivity of the nanocoral by measuring its ability to detect a standard chemical compound for Raman spectroscopy.
To get the nanocoral to target specific cells, the researchers took advantage of the capability to attach antibodies to polymer surfaces.
“We can tailor the nanocoral to cancer cells of interest by attaching the appropriate antibodies,” said the study’s other co-lead author, Liz Wu, who conducted this research as a Ph.D. student in the Applied Science and Technology program.
The researchers demonstrated this concept by coating the polystyrene surface with antibodies that attack human epidermal growth factor receptor 2 (HER-2), a well-known target for cancer treatment since it is often over-expressed in aggressive forms of breast cancer. They confirmed with both bright field and fluorescent images that the nanocoral attached to breast cancer cells with HER-2 receptors, while control experiments showed that no binding occurred when different antibodies or when cells lacking HER-2 were used.
“We are still in the early stages of development, but we are optimistic that the nanocorals will eventually become useful diagnostic and treatment tools for a wide range of cancers,” said Lee. “This will potentially allow us not only to deliver a drug, but also to see the response in real time at a sub-cellular level.”
Another co-author of the study is SoonGweon Hong, UC Berkeley graduate student in bioengineering.
The National Institutes of Health Nanomedicine Development Center for the Optical Control of Biological Function and the Defense Advanced Research Projects Agency helped support this research.