Are cancers newly evolved species?
Molecular biologist Peter Duesberg's theory that cancer results from chromosome disruption rather than a few gene mutations has led him to propose that cancers are actually evolving into autonomous species, akin to parasites.
July 26, 2011
Cancer patients may view their tumors as parasites taking over their bodies, but this is more than a metaphor for Peter Duesberg, a molecular and cell biology professor at the University of California, Berkeley.
Cancerous tumors are parasitic organisms, he said. Each one is a new species that, like most parasites, depends on its host for food, but otherwise operates independently and often to the detriment of its host.
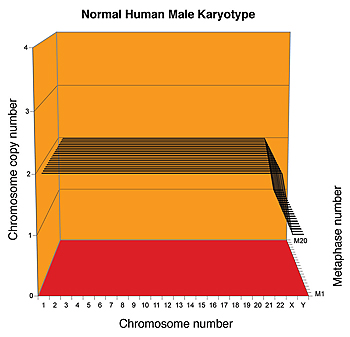
A karyograph is one way to display the number of copies of each chromosome in a clone of cells from an individual or a cancer. Here, the karyograph shows the chromosomes of 20 individual cells (represented by black lines) of a normal human male. Each cell has precisely two copies of 22 chromosomes and one copy of each sex chromosome, demonstrating that human cells have a fixed and stable karyotype.
In a paper published in the July 1 issue of the journal Cell Cycle, Duesberg and UC Berkeley colleagues describe their theory that carcinogenesis – the generation of cancer – is just another form of speciation, the evolution of new species.
“Cancer is comparable to a bacterial level of complexity, but still autonomous, that is, it doesn’t depend on other cells for survival; it doesn’t follow orders like other cells in the body, and it can grow where, when and how it likes,” said Duesberg. “That’s what species are all about.”
This novel view of cancer could yield new insights into the growth and metastasis of cancer, Duesberg said, and perhaps new approaches to therapy or new drug targets. In addition, because the disrupted chromosomes of newly evolved cancers are visible in a microscope, it may be possible to detect cancers earlier, much as today’s Pap smear relies on changes in the shapes of cervical cells as an indication of chromosomal problems that could lead to cervical cancer.
Carcinogenesis and evolution
The idea that cancer formation is akin to the evolution of a new species is not new, with various biologists hinting at it in the late 20th century. Evolutionary biologist Julian S. Huxley wrote in 1956 that “Once the neoplastic process has crossed the threshold of autonomy, the resultant tumor can be logically regarded as a new biologic species ….”
Last year, Dr. Mark Vincent of the London Regional Cancer Program and University of Western Ontario argued in the journal Evolution that carcinogenesis and the clonal evolution of cancer cells are speciation events in the strict Darwinian sense.
The evolution of cancer “seems to be different from the evolution of a grasshopper, for instance, in part because the cancer genome is not a stable genome like that of other species. The challenging question is, what has it become?” Vincent said in an interview. “Duesberg’s argument from karyotype is different from my argument from the definition of a species, but it is consistent.”
Vincent noted that there are three known transmissible cancers, including devil facial tumor disease, a “parasitic cancer” that attacks and kills Tasmanian devils. It is transmitted from one animal to another by a whole cancer cell. A similar parasitic cancer, canine transmissible venereal tumor, is transmitted between dogs via a single cancer cell that has a genome dating from the time when dogs were first domesticated. A third transmissible cancer was found in hamsters.
“Cancer has become a successful parasite,” Vincent said.
Mutation theory vs. aneuploidy
Duesbeg’s arguments derive from his controversial proposal that the reigning theory of cancer – that tumors begin when a handful of mutated genes send a cell into uncontrolled growth – is wrong. He argues, instead, that carcinogenesis is initiated by a disruption of the chromosomes, which leads to duplicates, deletions, breaks and other chromosomal damage that alter the balance of tens of thousands of genes. The result is a cell with totally new traits – that is, a new phenotype.
“I think Duesberg is correct by criticizing mutation theory, which sustains a billion-dollar drug industry focused on blocking these mutations,” said Vincent, a medical oncologist. “Yet very, very few cancers have been cured by targeted drug therapy, and even if a drug helps a patient survive six or nine more months, cancer cells often find a way around it.”
Chromosomal disruption, called aneuploidy, is known to cause disease. Down syndrome, for example, is caused by a third copy of chromosome 21, one of the 23 pairs of human chromosomes. All cancer cells are aneuploid, Duesberg said, though proponents of the mutation theory of cancer argue that this is a consequence of cancer, not the cause.
Key to Duesberg’s theory is that some initial chromosomal mutation – perhaps impairing the machinery that duplicates or segregates chromosomes in preparation for cell division – screws up a cell’s chromosomes, breaking some or making extra copies of others. Normally this would be a death sentence for a cell, but in rare cases, he said, such disrupted chromosomes might be able to divide further, perpetuating and compounding the damage. Over decades, continued cell division would produce many unviable cells as well as a few still able to divide autonomously and seed cancer.
Duesberg asserts that cancers are new species because those viable enough to continue dividing develop relatively stable chromosome patterns, called karyotypes, distinct from the chromosome pattern of their human host. While all known organisms today have stable karyotypes, with all cells containing precisely two or four copies of each chromosome, cancers exhibit a more flexible and unpredictable karyotype, including not only intact chromosomes from the host, but also partial, truncated and mere stumps of chromosomes.
“If humans changed their karyotype – the number and arrangement of chromosomes – we would either die or be unable to mate, or in very rare cases become another species,” Duesberg said. But cancer cells just divide and make more of themselves. They don’t have to worry about reproduction, which is sensitive to chromosomal balance. In fact, as long as the genes for mitosis are still intact, a cancer cell can survive with many disrupted and unbalanced chromosomes, such as those found in an aneuploid cell, he said.
The karyotype does change as a cancer cell divides, because the chromosomes are disrupted and thus don’t copy perfectly. But the karyotype is “only flexible within a certain margin,” Duesberg said. “Within these margins it remains stable, despite its flexibility.”
Karyographs display karyotype variability
Duesberg and his colleagues developed karyographs as a way to display the aneuploid nature of a cell’s karyotype and its stability across numerous cell cultures. Using these karyographs, he and his colleagues analyzed several cancers, clearly demonstrating that the karyotype is amazingly similar in all cells of a specific cancer line, yet totally different from the karyotypes of other cancers and even the same type of cancer from a different patient.
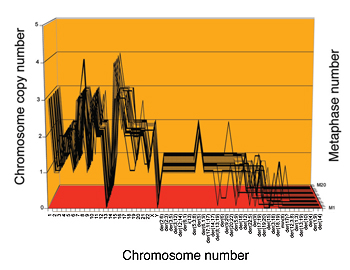
In contrast to normal cells, cervical cancer cells (HeLa) have flexible chromosomes. The 23 normal chromosomes have between 0 and 4 copies, while the several dozen hybrid or “marker” chromosomes have between 0 and 2. The copy numbers differ in the 20 individual HeLa cells shown, but they are nearly clonal, varying around an average clonal number.
HeLa cells are a perfect example. Perhaps the most famous cancer cell line in history, HeLa cells were obtained in 1951 from a cervical cancer that eventually killed a young black woman named Henrietta Lacks. The 60-year-old cell line derived from her cancer has a relatively stable karyotype that keeps it alive through division after division.
“Once a cell has crossed that barrier of autonomy, it’s a new species,” Duesberg said. “HeLa cells have evolved in the laboratory and are now even more stable than they probably were when they first arose.”
The individualized karyotypes of cancers resemble the distinct karyotypes of different species,, Duesberg said. While biologists have not characterized the karyotypes of most species, no two species are known that have the same number and arrangement of chromosomes, including those of, for example, gorillas and humans, who share 99 percent of their genes.
Duesberg argues that his speciation theory explains cancer’s autonomy, immortality and flexible, but relatively stable, karyotype. It also explains the long latency period between initial aneuploidization and full blown cancer, because there is such a low probability of evolving an autonomous karyotype.
“You start with a chromosomal mutation, that is, aneuploidy perhaps from X-rays or cigarettes or radiation, that destabilizes and eventually changes your karyotype or renders it non-viable,” he said. “The rare viable aneuploidies of cancers are, in effect, the karyotypes of new species.”
Duesberg hopes that the carcinogenesis-equals-speciation theory will spur new approaches to diagnosing and treating cancer. Vincent, for example, suspects that cancers are operating right at the edge of survivability, maintaining genomic flexibility while retaining the ability to divide forever. Driving them to evolve even faster, he said, “might push them over the edge.”
Duesberg’s colleagues are postdoctoral fellow Daniele Mandrioli and research associate Amanda McCormack of UC Berkeley and graduate student Joshua M. Nicholson in the Department of Biological Sciences at Virginia Polytechnic Institute.
Duesberg’s research is funded by the Abraham J. and Phyllis Katz Foundation, philanthropists Dr. Christian Fiala, Rajeev and Christine Joshi, Robert Leppo and Peter Rozsa of the Taubert Memorial Foundation, other private sources and the Forschungsfonds der Fakultät für Klinische Medizin Mannheim der Universität Heidelberg.
For more information:
- Is carcinogenesis a form of speciation? (Cell Cycle, Vol 10, Issue 13)
- Drug resistance argues against mutation theory of cancer (6/26/07 press release)
- UC Berkeley biologist disputes current dogma that genetic mutation is the cause of cancer (4/5/00 press release)
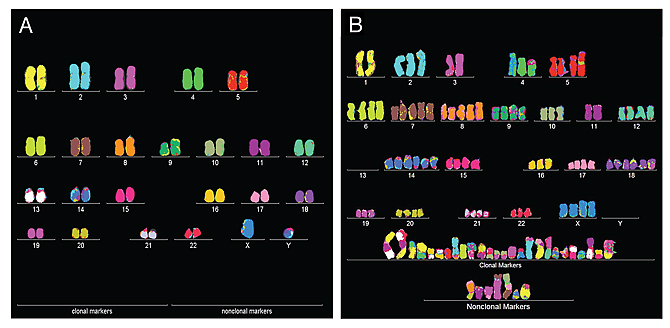
Staining chromosomes with different dyes highlights the orderly nature of the normal human karyotype (left), that is, humans have precisely two copies of each chromosome with no leftovers. A bladder cancer cell (right) has extra copies of some chromosomes, a few missing normal chromsomes, and a lot of hybrid or marker chromosomes, which characterize cancer cells.