Shining light on brain circuits to study learning, memory
NSF has given Ehud Isacoff, director of the Helen Wills Neuroscience Institute, $300,000 over two years to develop a new technology that uses light to tweak the synapses of brain cells to determine how they process information during learning and memory.

September 5, 2014
UC Berkeley neuroscientists plan to use light to tweak the transmission of signals in the brain to learn more about how the mouse brain and presumably the human brain process information.
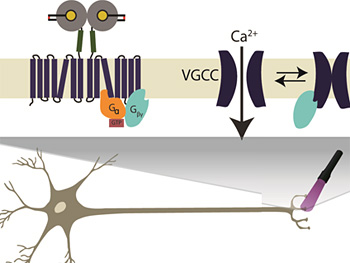
Inserting a light-sensitive switch (top left) into a glutamate receptor allows researchers to use violet light to turn on the receptor in presynaptic nerve terminals (bottom). This suppresses transmitter release and results in a smaller postsynaptic signal. Graphic by Ehud Isacoff, UC Berkeley.
Last month, the research project was awarded one of 36 new $300,000, two-year grants from the National Science Foundation in support of President Obama’s BRAIN Initiative, a multi-agency research effort that seeks to accelerate the development of new neuro-technologies that promise to help researchers answer fundamental questions about how the brain works.
NSF’s Early Concept Grants for Exploratory Research (EAGER) are designed to “enable new technologies to better understand how complex behaviors emerge from the activity of brain circuits,” according to an Aug. 18 announcement by the agency.
The new technology involves optogenetics, “an incredibly powerful approach to do circuit breaking to understand how the brain works,” said lead researcher Ehud Isacoff, UC Berkeley professor of molecular and cell biology and director of the Helen Wills Neuroscience Institute. “This is a way of exploring the basic principles of brain processing, and how these govern learning and memory.”
“And because these circuits play roles in anxiety, depression and schizophrenia, we can help define what goes wrong with brain circuits in disease,” he added.
Innovative optogenetic technology
Isacoff said that there is the critical need for new technologies that can record the activity of thousands of neurons at once, rather than the few now possible with standard electrophysiological recording. Optogenetics complements cell recording with the ability to control the activity of nerve cells. UC Berkeley’s optogenetic technique “goes a step further by controlling the way the cells communicate in the network.”
About a decade ago, Isacoff and UC Berkeley scientists Richard Kramer and Dirk Trauner – now at the University of Munich – developed ways to chemically synthesize a light-sensitive switch and attach it to signaling proteins on cell membranes so that a flash of light could turn the cells on and off. This allowed researchers to explore the function of the brain’s ion channels and the connectivity of neural circuits, offering the possibility of restoring sight to those with blinding retinal diseases such as retinitis pigmentosa.
Recently, Isacoff, Trauner and colleagues extended this optogenetic technique to the largest family of membrane receptors, which are involved in complex signaling not only in the brain but throughout the body. The ability to switch these G-protein coupled receptors (GPCRs) on and off with light now allows neuroscientists to manipulate fundamental modulatory functions of the synapse, the critical junction between nerve cells where signals are transmitted.
“Now we can directly probe the synapse, turning on and off the very receptors that mediate information filtering and adjust the strength of synapses,” he said. “The NSF-EAGER grant and a linked grant from the U.S.-Israel Binational Science Foundation to collaborator Eitan Reuveny at the Weizmann Institute enable us to take the next step: to generate a new family of light-controlled GPCRs that will make it possible to probe one of the central mechanisms of learning and memory in motor control.”
For more information on the projects funded by NSF, link to the EAGER website.
RELATED INFORMATION
- Chemical makes blind mice see (2012)
- Photoswitches shed light on burst swimming in zebrafish (2009)
- Photoswitches could restore sight to blind retinas (2006)